Changing the Microstructure by Heat Treatment
The properties of steel are not necessarily optimal after production. The most important properties of a steel grade are defined by the alloys. However, some characteristics can be further improved using heat treatment.
There are many methods of heat treatment. Some change the structure of the entire cross section; others only affect the surface layer and have little or no influence on the inside. Characteristics that can be manipulated by heat treatment include hardness, tensile strength and toughness.
The Temperature-time Sequence
Heat treatment cycle consists of three steps: heating, holding and cooling. To achieve specific characteristics, it is important to adhere exactly to a predetermined temperature-time sequence. The iron-carbon diagram shows the microstructural change during infinitely slow cooling. When the cooling speed changes however, other microstructures may form.
The structural change during heating can be read from the time-temperature-austenitization diagram. The diagram is only valid for a very specific steel grade because the microstructure strongly depends on the alloy. To achieve good hardening results it is favourable to heat to a temperature at which homogenous austenite is formed. Homogenous austenite is able to dissolve the carbon and incorporate it into the atomic lattice. When this temperature is reached in the surface layer it must be held untilthe entire volume reaches the same temperature. Additional holding time must be added until the transformation reaches the desired state. Depending on the intended use, incomplete conversion may also be desirable.
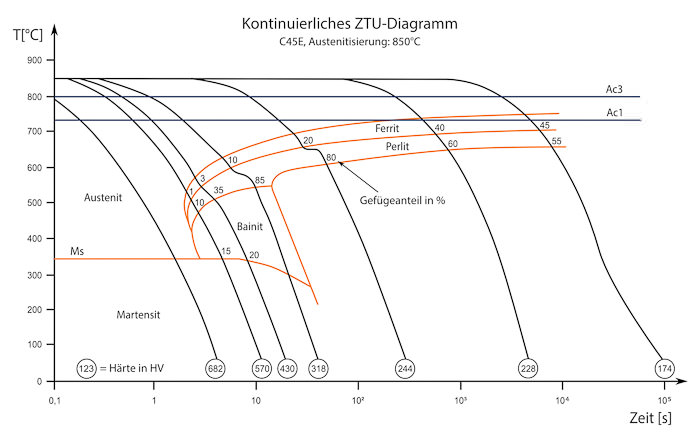
During the cooling phase different microstructural transformations take place. These are shown in the isothermal transformation diagram. When the material is cooled quickly, the transformation of Austenite to perlite occurs quickly at around 723°C. The carbon is not able to diffuse and the transformation and exsolution into cementite is impeded. When cooling very quickly with a gradient of over 15°C/s bainite is formed instead of perlite. In bainite the atomic lattice is distorted by embedded carbon.
At extremely fast cooling speeds the transformation does not occur until reaching a temperature of 400°C. When the transformation occurs a space-centred tetragonal lattice is formed. This crystal structure is called martensite and is very hard and brittle.
Methods and their Purposes
To harden steel, it is necessary to achieve a high proportion of martensite. This requires a very steep cooling gradient; the process is called quenching. However, depending on the alloy some residual bainite may exist.
Hardening creates high tensions and a very brittle material. Often it is therefore necessary to temper the material by re-heating it to a lower temperature. This allows the transformation of some of the martensite and makes it possible to fine tune malleability and strength.
Austempering is a process to create a bainitic steel. Typically it does not reach the hardness of martensite.
For case hardening only the surface layer of the material is heated. Therefore transformations only take place near the surface and the volume is not affected, otherwise they are equivalent to the transformations during hardening. Heating can be achieved using induction.
Other hardning methods include carburizing and nitriding, where the steel is heated, packed in carbon or surrounded by nitrogen to increase the carbon content in the surface layer or to form nitrides. Both processes create an extremely hard surface.
Soft-annealing, stress-relief annealing, normalizing, coarse-grain annealing, homogenization and recristallization annealing take place at lower holding temperatures and slow cooling gradients. All of these methods have the common goals of changing the granulation or releasing stress.